Enemeez 283mg Docusate Sodium Mini Enemas/Stool Softeners - Constipation Relief for Adults, 35 Count
 Limited Time Sale
Limited Time Sale$47.39 cheaper than the new price!!
Free cash-on-delivery fees for purchases over $99
Product details
| Management number | 219373348 | Release Date | 2026/05/03 | List Price | $31.60 | Model Number | 219373348 | ||
|---|---|---|---|---|---|---|---|---|---|
| Category | |||||||||
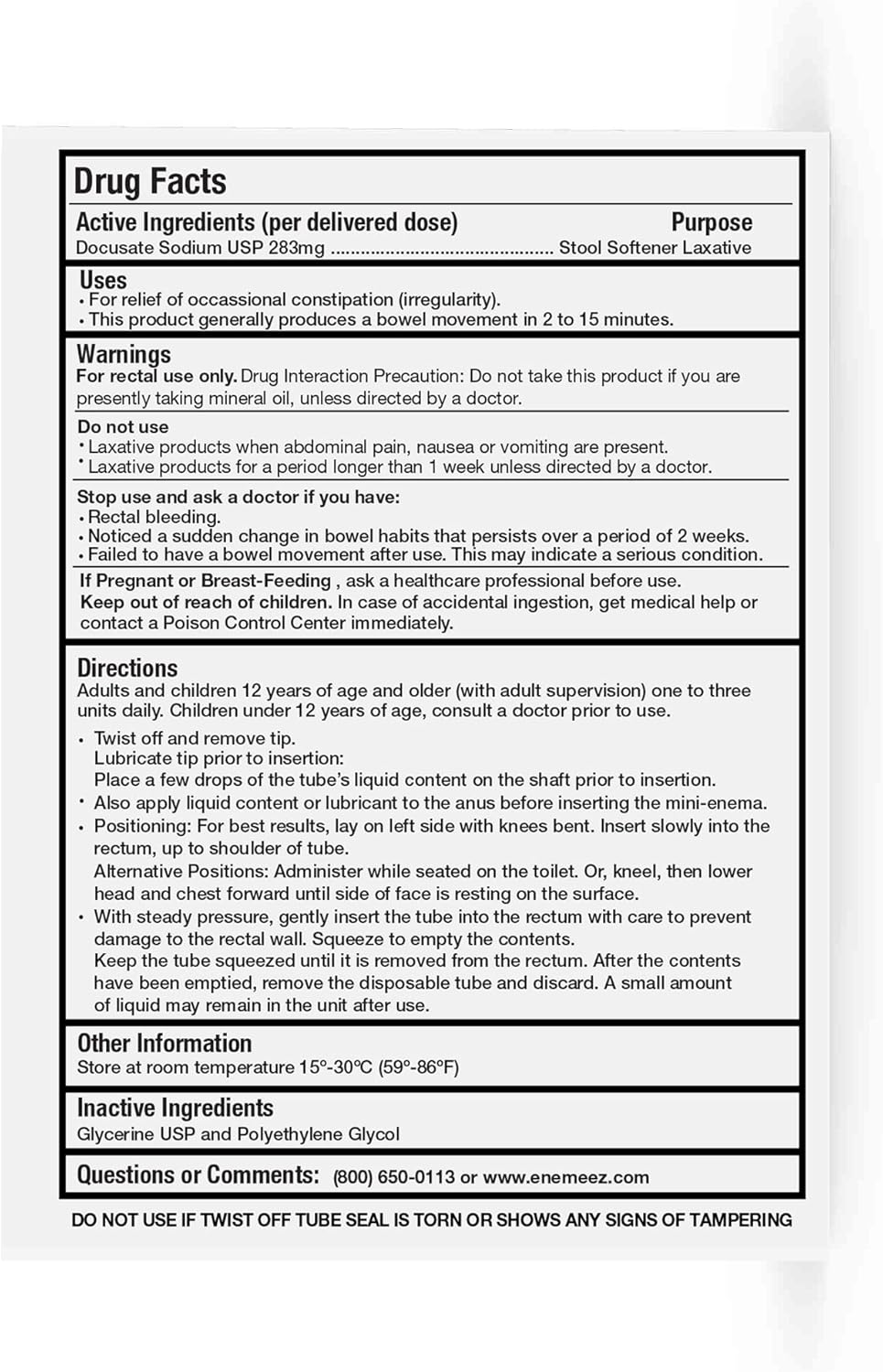
Enemeez Regular 283mg Docusate Sodium Mini Enema offers a fast-acting solution for constipation relief. With its formulation, it delivers results in just 2 to 15 minutes, making it one of the fastest options available. The active ingredient, docusate sodium, works by drawing water into the bowel, softening the stool, and promoting bowel movement. This hyper-osmotic laxative stimulates nerve endings in the bowel lining, triggering peristalsis for bowel evacuation. Designed with a comfortable, flexible tip, it ensures ease of use for home applications. Enemeez mini-enema is formulated to provide reliable relief without causing irritation, addressing constipation commonly encountered in individuals with spinal cord injuries, Spina Bifida, Multiple Sclerosis, and Traumatic Brain Injury. By supporting a systematic bowel care program, it helps prevent health complications and enhances quality of life. The non-irritating properties make it safe, offering an alternative to oral laxatives that may require hours or days to take effect. For those seeking targeted, rapid relief, Enemeez delivers a good solution.
- FAST RELIEF: Experience fast-acting constipation relief with Enemeez's formula, ideal as laxatives for constipation relief within minutes.
- SAFE FORMULATION: The non-irritating formula offers safe and effective laxatives for constipation relief for adults
- EASY TO USE: Designed for home use, these enemas come with a flexible tip for comfortable insertion, making constipation relief easy.
- SOFTENS STOOL: With the active ingredient docusate sodium, an osmotic laxative, our enema softens stool by mixing it with watery fluids to aid severe constipation relief and improve bowel movements
- FOR RECTAL USE: Rectal application only. Before using one of the enemas, put a drop of liquid on the enema shaft, perfect for constipation relief for men and women
| UPC | 317433000409 |
|---|---|
| Item Form | Drop |
| Brand Name | Enemeez Plus |
| Unit Count | 35 Fluid Ounces |
| Manufacturer | Enemeez |
| Model Number | ENE00040 |
| Container Type | Tube |
| Item Dimensions | 6.5 x 5.25 x 2.25 inches |
| Number of Items | 1 |
| Target Audience | Adults and children 12 years of age and up |
| Product Benefits | Constipation Control |
| Package Type Name | Tube |
| Active Ingredients | Docusate Sodium |
| Additional Features | Non-irritating formula |
| Age Range Description | Adult |
| Manufacturer Part Number | ENE00040 |
| Specific Uses For Product | Digestion |
Correction of product information
If you notice any omissions or errors in the product information on this page, please use the correction request form below.
Correction Request Form